Science & Technology
Biofilm Formation in Bacteria: How Do Microbial Communication Alters Bacterial Behavior and Antibiotic Resistance
By Chung, Hyeseung Leah
March 23, 2026
Images
Article
Introduction
We, the humans, are known to be social animals, who rely on community corporations, social connections, and community support for survival and security. However, it is not only us that live in a community. Very tiny organisms, which are so small that you are not able to see with your bare eyes, form a community for their survival too! These organisms are called bacteria. Although bacteria are known to be unicellular organisms, they typically do not exist as isolated cells (also called planktonic bacteria) in a natural habitat. Planktonic bacteria who exist as individual cells that move independently with a fast growth rate and less communication are in the “dispersal” phase, rather than primarily the lifestyle of bacteria. Instead, they often form biofilms, which are structured communities of bacteria that stick to each other and to a surface. Biofilms can be found in diverse environments: a dental plaque on human teeth, water environments, and even in medical devices, sometimes leading to an infection. The shift from planktonic bacteria to a biofilm significantly changes bacterial behavior and resistance to environmental change. This transition from harmless planktonic bacteria to pathogenic biofilm communities explains why some infections become chronic and difficult to treat. The biofilm shows that cooperation is a powerful survival strategy across life forms, from microorganisms to human societies, but understanding how bacteria cooperate and communicate also can help scientists to develop better treatments for infections.
Stages of Biofilm Formation
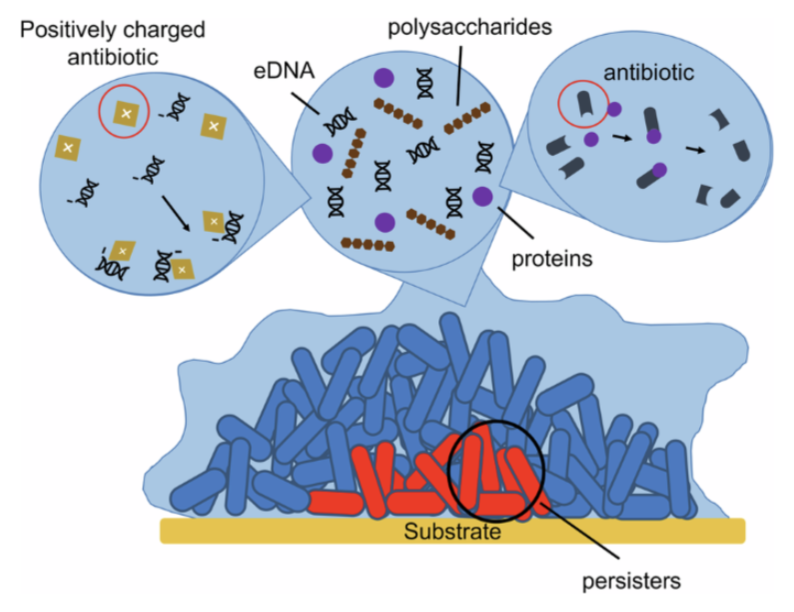
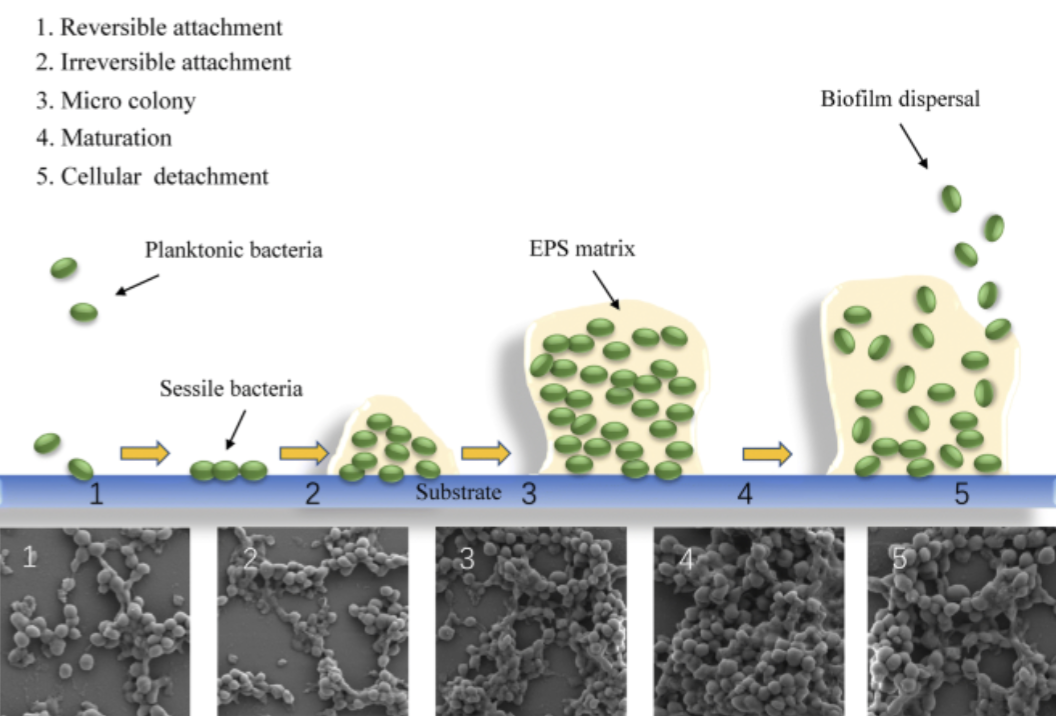
Biofilm formation has five main stages: reversible attachment, irreversible attachment, maturation I, maturation II, and dispersion. During the reversible attachment, planktonic bacteria attach to a surface using flagella for movement, and pili or fimbriae for temporary attachment. This attachment is weak and bacteria can still detach from the surface easily. The initial step is affected by several environmental factors such as temperature, nutrient availability, and surface properties. The second stage, which is irreversible attachment, is when bacteria begin producing extracellular polymeric substances (EPS), a matrix composed of polysaccharides, proteins, and extracellular DNA. EPS acts like a glue, sticking the cells permanently to the surface. At this stage, bacteria start to divide and form microcolonies. The third and fourth stages, maturation I and maturation II, involve development of a fully structured biofilm. The microcolonies grow into 3D structures covered by a thick EPS matrix. Water channels are formed within the structure for the nutrient and oxygen circulation and waste removal. As a biofilm maturation process, cells differentiate depending on their position in biofilm. Cells in the outer location of biofilm are often more active due to more oxygen and nutrients contacting them, while inner cells often enter dormant states or have slow growth rate with limited nutrient exchange. Finally, when environmental signals such as nutrient limitation occur, some bacteria detach from the biofilm and will return to a planktonic state. This stage is called dispersion. These dispersed cells can even colonize new surfaces, continuing the biofilm life cycle in other places.
Antibiotic Resistance
Biofilms dramatically change the behavior of bacteria compared to planktonic cells. For example, increased antimicrobial resistance is one of the most significant changes. The EPS matrix that covers biofilm limits the antibiotics to penetrate into the cells, and reduced growth rate of the inner cells makes antibiotics less effective. In detail, biofilm contains persister cells—small subpopulations of bacteria that are in dormant states—surviving antibiotics without being genetically resistant to it because antibiotics usually target cell activities such as cell wall synthesis, DNA replication, protein production, or cell division. The study of antibiotic resistance says that bacteria in biofilms exhibit 10-1,000 times higher resistance than the planktonic cells. This antibiotic resistance can lead to infections being recurred, and harder to treat.
Communication and Division of Labor
Moreover, Biofilm bacteria communicate through quorum sensing, a process in which bacteria release or detect ligands (molecules). When the size of population reaches an optimal range, the signal can trigger gene expression regulation (turning genes on or off), biofilm regulation (activating genes that produce EPS), virulence genes activation (molecules that help causing disease), and metabolic activity. This communication is vital in biofilm, as it makes bacteria function as a coordinated community rather than individual cells. Furthermore, biofilm formation develops metabolic specialization and division of labor. Nutrient gradients within the biofilm create varied metabolic activities among cells, enhancing bacteria’s survival under harsh conditions. Streptomyces is an example of bacteria dividing roles in biofilm formation, which they focus on producing antibiotics, helping them to compete with other microbes, and growth and reproduction.
Treatments
Then what are some possible treatments for pathogens in bacterial biofilms? Usually pathogenic bacteria are treated with multiple antibiotics together, which penetrate the biofilm better. The treatment to kill pathogenic bacteria within biofilm should be prolonged and higher dosage, because biofilms are protected by the EPS matrix. However, there are MDR (multidrug-resistant) biofilms. For alternatives, scientists place antibiotics into nanoparticles that are made of lipids of fats (such as liposomes) which is then more able to penetrate into EPS matrix or targeting biofilm associated genes by using EPS-degrading enzymes (such as PslGh, DNase I, and protease) that break down the EPS matrix. Yet, antibiotic resistance is developing in many pathogens, and further treatments should be investigated with more knowledge in these microorganisms.
Conclusion
Overall, biofilm formation enhances survival capabilities by transforming planktonic bacteria into organized, coordinated communities. Through stages of reversible attachment, irreversible attachment, maturation I, maturation II, and dispersion, bacteria produce EPS matrix (which they change certain behaviors), communicate with quorum sensing, and develop into metabolic specialization. Understanding this is crucial for dealing with chronic infections and antibiotic resistance.
Citation
Dal Co, A., Brannon, C., & Ackermann, M. (2018). Division of labor in bacteria. eLife, 7, e38578. https://doi.org/10.7554/eLife.38578
Davey, M. E., & O'Toole, G. A. (2000). Microbial biofilms: from ecology to molecular genetics. Microbiology and molecular biology reviews : MMBR, 64(4), 847–867. https://doi.org/10.1128/MMBR.64.4.847-867.2000
Donlan R. M. (2002). Biofilms: microbial life on surfaces. Emerging infectious diseases, 8(9), 881–890. https://doi.org/10.3201/eid0809.020063
Hernández, A. (2025, February 4). Biofilms: What are they, formation, removal, and more. Osmosis. https://www.osmosis.org/answers/biofilm
Mishra, S., Gupta, A., Upadhye, V., Singh, S. C., Sinha, R. P., & Häder, D. P. (2023). Therapeutic Strategies against Biofilm Infections. Life (Basel, Switzerland), 13(1), 172. https://doi.org/10.3390/life13010172
Stewart, P. S., & Costerton, J. W. (2001). Antibiotic resistance of bacteria in biofilms. Lancet (London, England), 358(9276), 135–138. https://doi.org/10.1016/s0140-6736(01)05321-1